 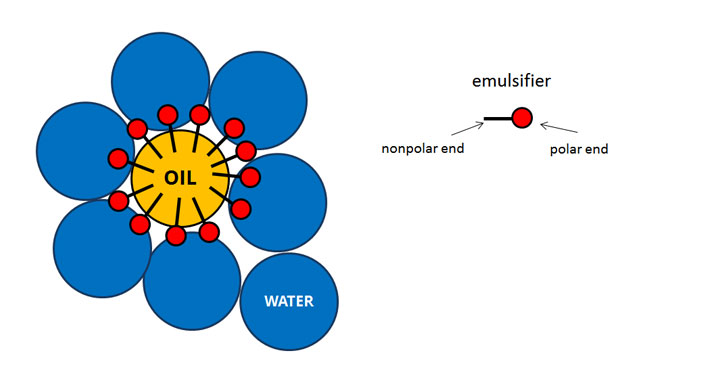
The term dispersion also refers to the physical property of the degree to which particles clump together into agglomerates or aggregates. However, stirring the mixture with a spoon will create turbulent flows in the water that accelerate the process of dispersion through convection-dominated dispersion. The molecules in a drop of food coloring added to water will eventually disperse throughout the entire medium, where the effects of molecular diffusion are more evident. These phenomena are reflected in common real-world events. Diffusion is the dominant mechanism in the process of dispersion in cases of little to no turbulence in the bulk, where molecular diffusion is able to facilitate dispersion over a long period of time. With respect to convection, variations in velocity between flow paths in the bulk facilitate the distribution of the dispersed material into the medium.Īlthough both transport phenomena contribute to the dispersion of a material into the bulk, the mechanism of dispersion is primarily driven by convection in cases where there is significant turbulent flow in the bulk. This unequal distribution results in a concentration gradient that drives the dispersion of particles in the medium so that the concentration is constant across the entire bulk. When the dispersed material is first introduced into the bulk medium, the region at which it is introduced then has a higher concentration of that material than any other point in the bulk. With respect to molecular diffusion, dispersion occurs as a result of an unequal concentration of the introduced material throughout the bulk medium. This process is facilitated by molecular diffusion and convection. Also, the sudden appearance of conductivity in a system of a dispersed conductive phase in an insulating matrix has been explained.ĭispersion is a process by which (in the case of solid dispersing in a liquid) agglomerated particles are separated from each other, and a new interface between the inner surface of the liquid dispersion medium and the surface of the dispersed particles is generated. This is the reason why some liquid dispersions turn to become gels or even solid at a concentration of a dispersed phase above a critical concentration (which is dependent on particle size and interfacial tension). Both surfaces have to be created (which requires a huge amount of energy), and the interfacial tension (difference of surface tension) is not compensating the energy input, if at all.Įxperimental evidence suggests dispersions have a structure very much different from any kind of statistical distribution (which would be characteristics for a system in thermodynamic equilibrium), but in contrast display structures similar to self-organisation, which can be described by non-equilibrium thermodynamics. To understand the formation and properties of such dispersions (incl emulsions), it must be considered that the dispersed phase exhibits a "surface", which is covered ("wet") by a different "surface" that, hence, are forming an interface (chemistry). In the following, only such dispersions with a dispersed phase diameter of less than 1 µm will be discussed. There are only very few studies about the structure of dispersions (emulsions), although they are plentiful in type and in use all over the world in innumerable applications (see below). 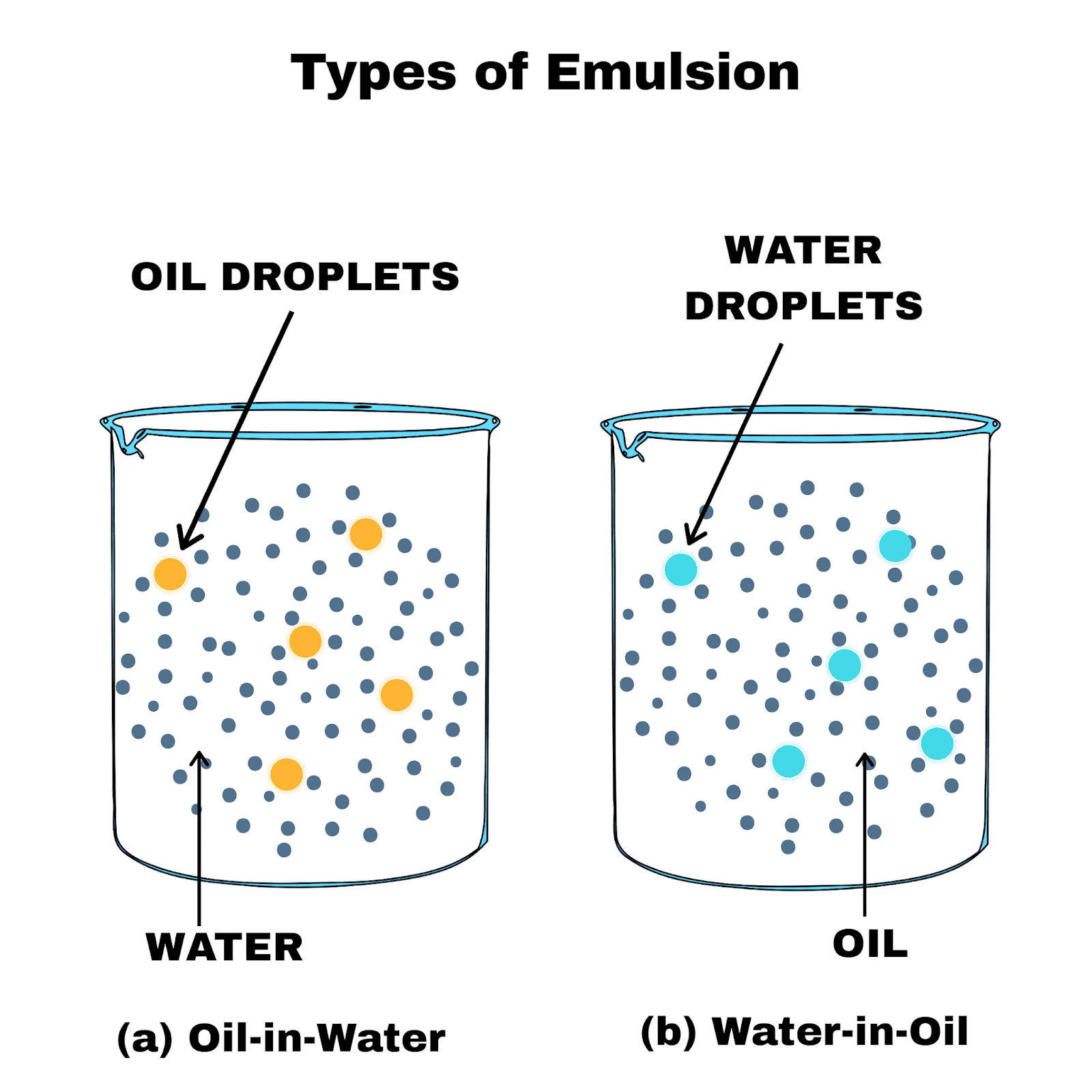
However, percolation theory can be applied only if the system it should describe is in or close to thermodynamic equilibrium. Therefore, for dispersions, usually percolation theory is assumed to appropriately describe their properties. In general, dispersions of particles sufficiently large for sedimentation are called suspensions, while those of smaller particles are called colloids and solutions.ĭispersions do not display any structure i.e., the particles (or in case of emulsions: droplets) dispersed in the liquid or solid matrix (the "dispersion medium") are assumed to be statistically distributed. The two phases may be in the same or different states of matter.ĭispersions are classified in a number of different ways, including how large the particles are in relation to the particles of the continuous phase, whether or not precipitation occurs, and the presence of Brownian motion. Ī dispersion is a system in which distributed particles of one material are dispersed in a continuous phase of another material. Note 1: Modification of definition in ref. Material comprising more than one phase where at least one of the phases consists of finely divided phase domains, often in the colloidal size range, dispersed throughout a continuous phase. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
